drug-development


The resolution revolution in cryo-electron microscopy had reached broad recognition by 2015, when Nature Methods hailed it the method of the year and declared an end to “blob-ology,” a term structural biologist Helen Saibil recalls crystallographers once used to dismiss EM’s then low-resolution imaging. In the decade since, pharma companies have moved from cautious exploration… The post Thermo Fi…

IntroductionVorolanib is a new-generation multi-targeted kinase inhibitor (MTKI) whose drug-drug interaction (DDI) profile remains largely unexplored. This study combined in vitro and in vivo approaches to systematically investigate the effect of resveratrol on vorolanib metabolism.MethodsUsing ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS), the concentrations of vo…
(WSJ) – Drug companies like Eli Lilly and Roche are racing to build supercomputers to help fix the 90% failure rate in drug development Drug companies have been talking about the potential for AI to supercharge drug development for years, … Read More

For faster development of drugs and knowledge: The PodoHealthX research team at Greifswald University Medicine has developed a method that uses 3D microscopy to provide fast and precise insights into kidney corpuscles. The method makes it possible to isolate animal kidney corpuscles in the shortest ...


Drug development has always required decisions to be made in the face of uncertainty. Which dose should move forward? Which patient population is most appropriate? How should a study be designed to generate meaningful evidence? For many years, those decisions have been shaped through a largely empirical and sequential process—where decisions are made primarily based on the data generated in the …

Parkinson’s disease is a condition that slowly affects the brain and makes movement harder over time. People with Parkinson’s may notice shaking, stiffness, and trouble with balance. These symptoms happen because certain brain cells stop working properly. These cells are responsible for making dopamine, a chemical that helps control smooth movement. Most current treatments help […] The post Exper…

BackgroundNarciclasine has demonstrated anticancer activity at low nanomolar concentrations in various preclinical cancer models, but no clinical data in cancer patients are available. Although its biological activity and structure-activity relationships are relatively well characterized, the pharmacological properties of narciclasine have not been reported. This information will benefit the rese…

Noncovalent PROTAC Antibody Drug Conjugates Krum, David Proteolysis Targeting Chimeras (PROTACs) are a new class of heterobifunctional drugs that induce proximity between an E3 ligase and a given protein of interest (POI), causing the POI to be ubiquitinated and targeted for proteasomal degradation. While the event-driven, irreversible pharmacodynamics of PROTACs offer advantages over traditional…

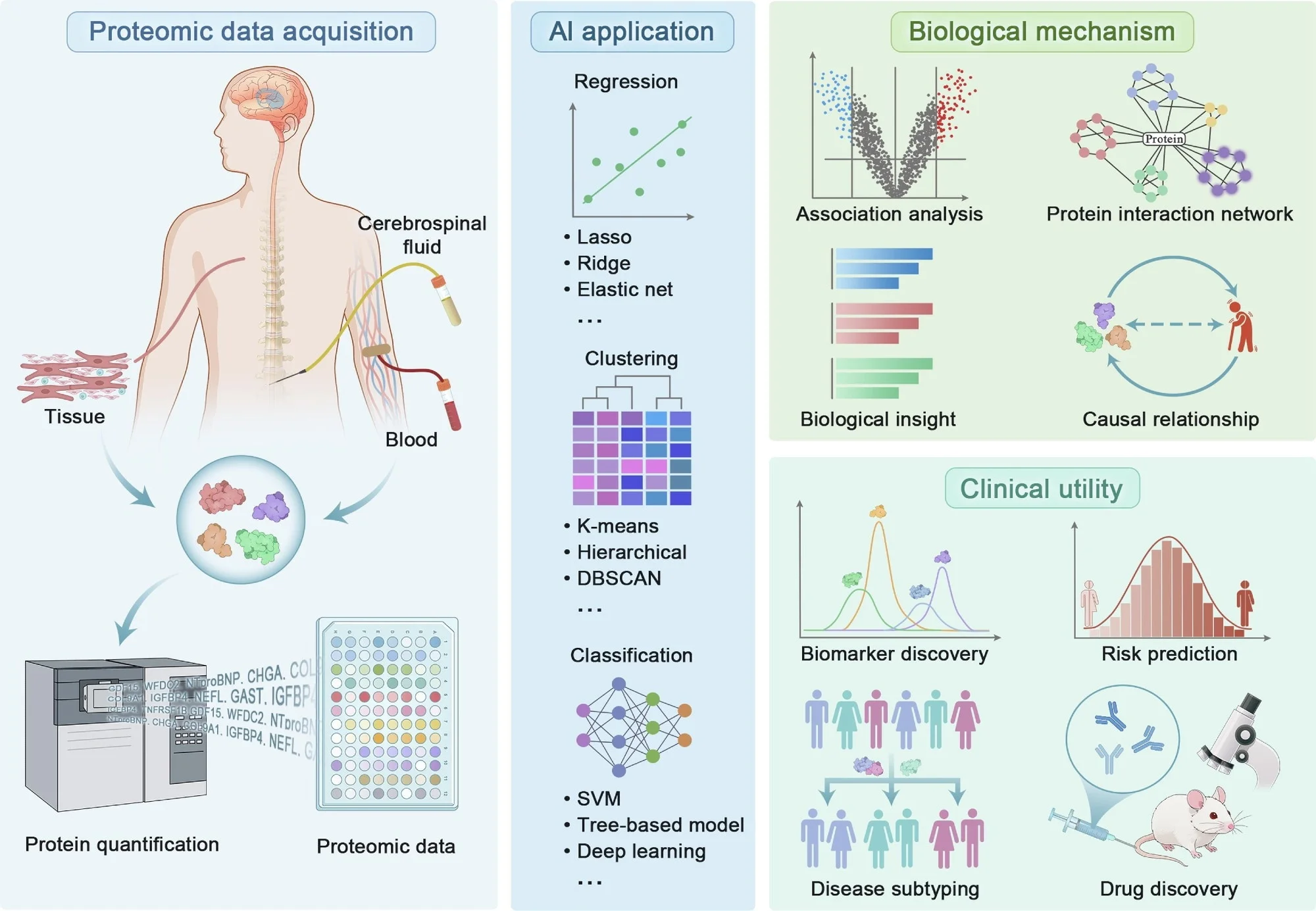
This review examines how high-throughput proteomics is expanding precision medicine by improving biomarker discovery, disease prediction, and drug development. It also shows how AI is helping researchers interpret complex proteomic data while highlighting major barriers such as standardization, validation, and clinical translation.

IntroductionZapnometinib (ATR-002) is a selective MEK inhibitor designed to modulate the MAPK/ERK pathway, which plays a key role in viral infections and inflammatory diseases. Clinical characterization of its safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) is essential to support further development. This Phase I clinical trial was registered under EudraCT number 2021-0052…


Clinical development is entering a period in which acceleration and accountability are advancing in parallel. Study teams are operating in an environment shaped by evolving regulatory expectations, expanding data sources, and rapid innovation in analytics and automation. As a result, the industry is aligning around shared priorities: greater visibility into trial execution, stronger cross-functio…

UVA’s Nikolay V. Dokholyan, PhD, and colleagues have developed a suite of artificial intelligence-powered tools, called YuelDesign, YuelPocket and YuelBond, that work together to transform how new drugs are created. The centerpiece, YuelDesign, uses a cutting-edge form of AI called diffusion models to design new drug molecules tailored to fit their protein targets exactly, even […] The post New A…


Recent regulatory developments are reshaping how sponsors think about evidence, oversight, and operational readiness. In a New England Journal of Medicine Sounding Board, FDA leadership announced that one adequate and well-controlled clinical trial — supported by confirmatory evidence — could become the agency’s new default standard for approval.i The shift formally moves away from the long-stand…


In February 2026, the Food and Drug Administration (FDA) outlined a meaningful evolution in the Agency’s evidentiary approach to drug approval: establishing one adequate and well-controlled trial, supported by confirmatory evidence, as the default basis for marketing authorization. This position was articulated in The New England Journal of Medicine in “One Pivotal Trial, the New... The post The …


Gaining regulatory approval for drugs targeting conditions that cannot ethically be studied in humans presents a unique challenge for drug developers. Without traditional clinical trial pathways, teams must rely on alternative methods to demonstrate efficacy and support approval. This webinar will explore how modeling and simulation are used to meet the requirements of the FDA […] The post Naviga…

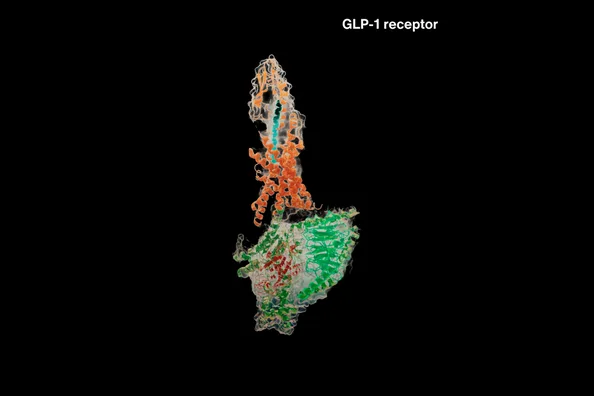
With near-atomic precision, a USC Dornsife team of scientists visualized for the first time how opioids such as loperamide and the antidote naloxone engage a key brain receptor, offering insight that could lead to better pain treatments and improved overdose reversal. The post A path to safer painkillers – revealed by freezing opioids and their protein receptors in motion appeared first on USC Do…


Seattle biotech companies are using AI to design new medical treatments, but scientists caution the tech holds enormous promise, but needs to stay grounded in reality. Read More
An EMBL alumnus’ work with tRNA, along with support from EMBLEM, has led to a start-up, Umlaut.bio, that is positioned to aid drug development.


Eyescreen, Inc. Receives SBIR Funding to Use Human Eyes for Testing of Eye Disease and Vision Loss, Reducing Reliance on Animal Models.
 research.io
research.ioSign up to keep scrolling
Create your feed subscriptions, save articles, keep scrolling.