Dr. Joaquin Barroso's Blog
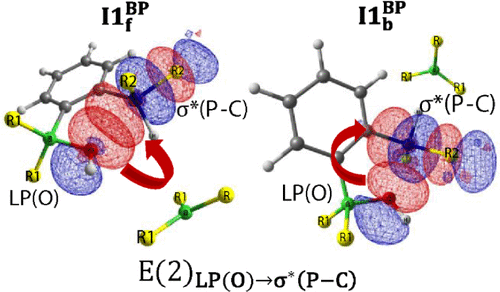
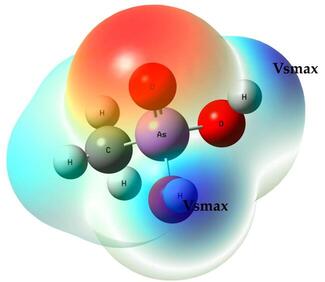
Obtaining hydrogen from water is one of those topics that attracts lots of attention from many fronts in chemistry; on paper, it can be clean, elegant, and deceptively simple. In practice, however, water is stubborn and breaking it apart requires catalysts, large energy inputs, or clever chemistry that juggles thermodynamics and kinetics. Among the many clever ideas proposed over the last decade,…
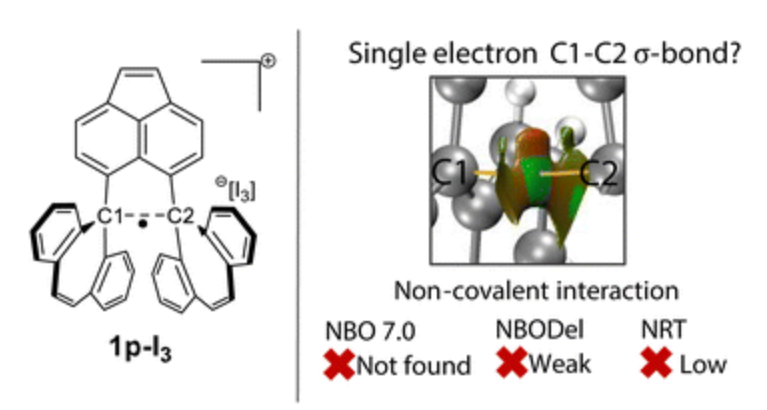
The compound spiro-dibenzocycloheptatriene (figure 1) made headlines a year ago by claiming that the ipso carbon atoms across the molecule were bound together by a single electron σ bond. This is truly a seductive idea: that somewhere out there, in the shadows of open-shell aromatics, carbon atoms might form a single-electron σ-bond — a unicorn of bonding, indeed. Since bold claims require bold s…
The strength of a chemical bond can be defined as the change in enthalpy when a bond is homolytically broken into two radicals. Bond dissociation energy is thus the measure of the strength of a bond and, while it is temperature dependent, it can be calculated by DFT or ab initio methods. In this tutorial we’re going to calculate the C-H bond dissociation energy (BDE) in the methane (CH4) molecule…

2025 was declared by UNESCO as the International Year of Quantum Sciences, bringing a lot of celebrations, however much of the discourse is dominated by physicists and quantum technologists, some of which proclaim the dawn of the new “quantum revolution”. Yet, amid the excitement, I can’t help but notice that quantum chemistry remains underrepresented, despite being the discipline that has arguab…
Unrecognized potential number 6 in GetPot A rather silly error in Gaussian, but one that seems to be common and to which I haven’t found any posted solutions, so here goes mine. This error shows up when reading an external basis set and looks like this (color emphasis mine): Rotational constants (GHZ): 0.0492351 0.0073969 0.0065324 Leave Link 202 at Tue Sep 30 16:14:43 2025, MaxMem= 104857600 cpu…
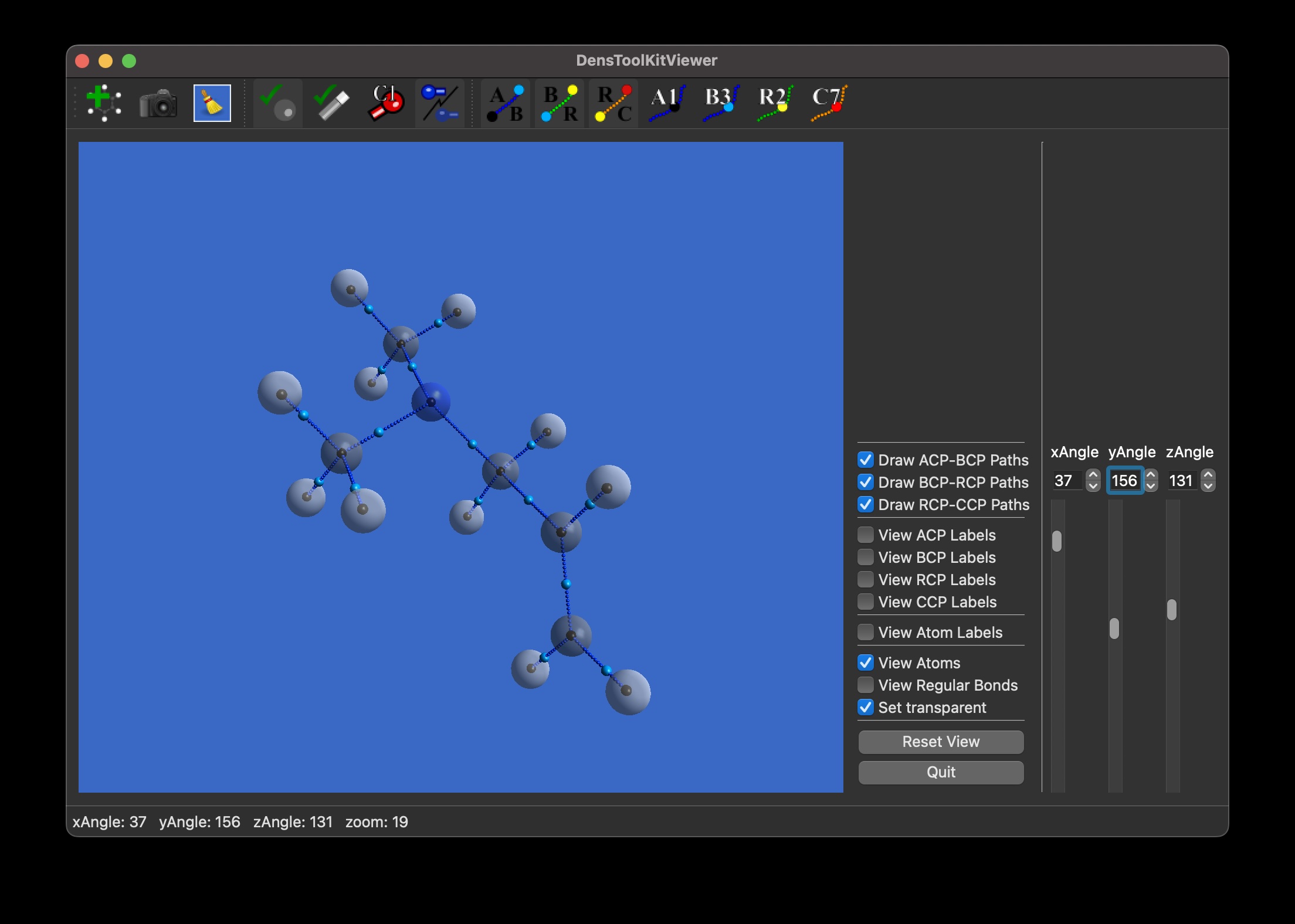
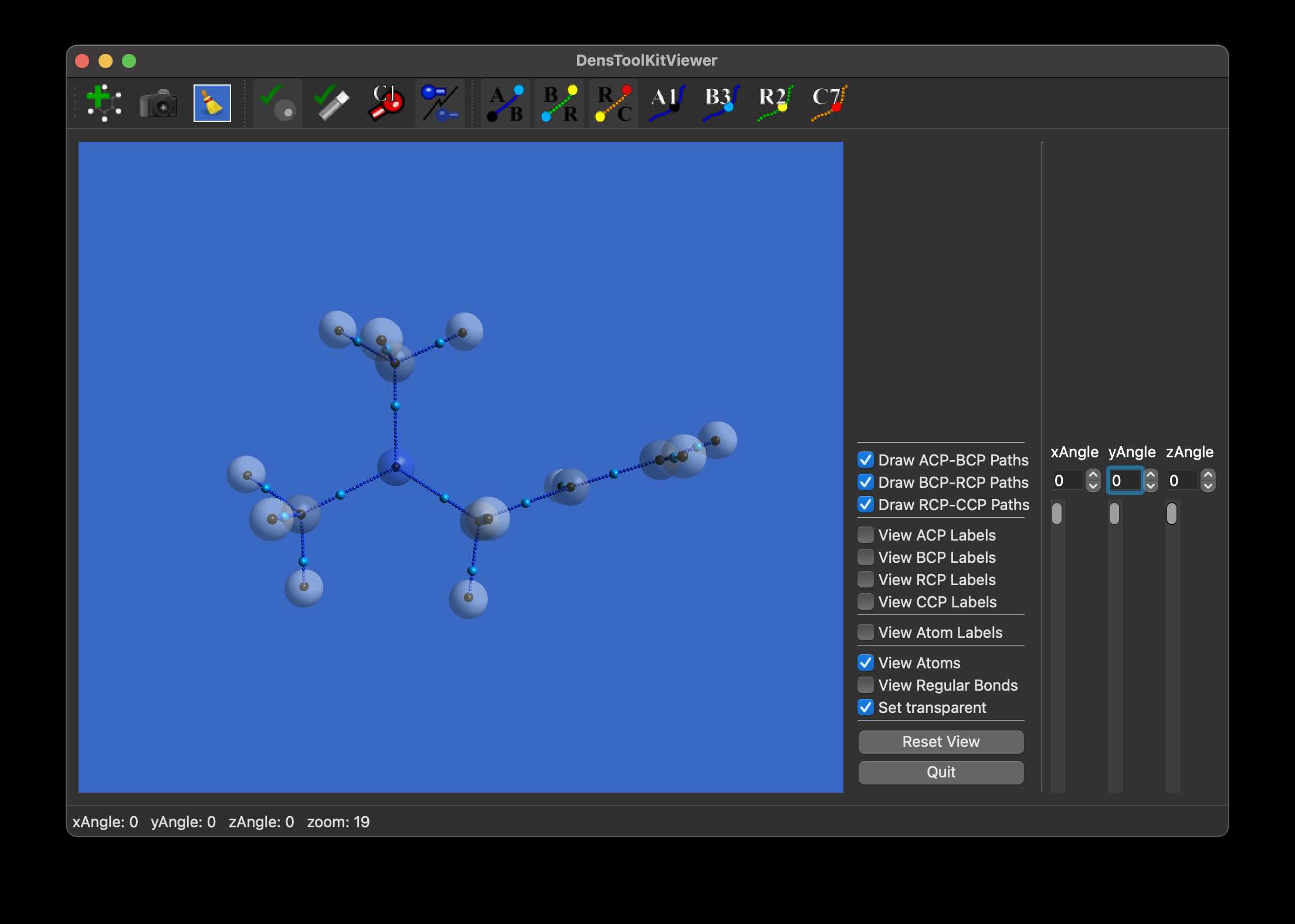
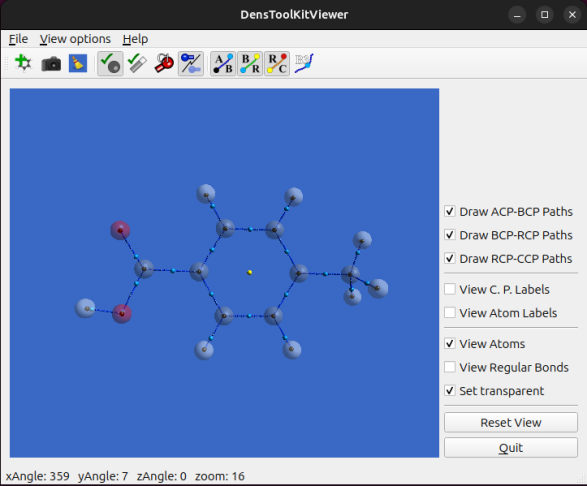
In this tutorial, we shall prepare a figure using DensToolKitViewer 2.1.0 (or later) to select the viewing angle of the molecule, for whenever the default view isn’t the optimal viewpoint or to get various for comparison. Installation and main controls were already covered in a previous post, as well as an update for ease of use. We use the file N-N-dimethyl-Propen-1-amine.wfx which is located in…
The new DensToolKitViewer just got even better! So, if you want to update DTKViewer to make the most of the new features, in this tutorial we shall compile and install DensToolKitViewer. We provide the explicit steps to compile under Ubuntu and under MacOSX. We will assume that the git repo is cloned in /home/your_username/Downloads . For this, execute the following commands $ cd ~/Downloads $ gi…
Preparing publication quality images from your calculations is essential. To pre-visualize images to get the best orientations of your molecule, DTK2.0 includes two options: a new GUI called DensToolKitViewer and DTKQDMol. To install DensToolKitViewer, you need CMake and the QT-5 libraries all of which you can install in the following way (Ubuntu): ~$sudo apt install -y qtbase5-dev qt5-qmake cmak…
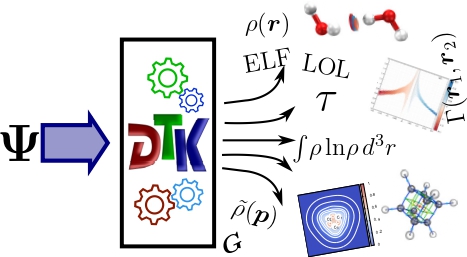
DensToolKit2 is a versatile open-source, cross-platform, suite of programs for analyzing electronic densities calculated from various sources and getting cool plots for some of the most popular scalar fields out there, such as electron densities, spin densities, visualizing critical points, or investigating complex topologies of noncovalent interactions, DensToolKit2 provides an integrated, scrip…
Thanks to all for reading! This Blog has been running for free since 2009 and will continue to do so. If you’ve found it useful or would like to support our efforts, consider making a donation. Your support is truly appreciated! To make a donation, please visit paypal.me/joaquinbarroso1
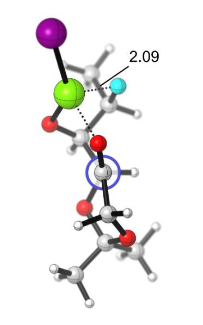
The Grignard addition is one of those reactions you learn during your first year of Organic chemistry and probably the last you forget when you become a computational chemist. It was Victor Grignard who became one of the earliest Nobel Laureate in Chemistry ever, and I know it took me a while to recognize it […]

Computational chemistry is a field that thrives on innovation, precision, and the ability to tackle complex problems. With the rise of artificial intelligence (AI) and machine learning (ML), tools like DeepSeek are becoming indispensable for researchers and students alike. In this post, we’ll explore how DeepSeek can help computational chemists advance their research, how it […]
Calculating reaction constants, in the correct physical way, requires accurate thermodynamic calculations involving solvation/desolvation energies at high levels of theory, because free energy deviations lower than 1 kcal/mol lead to reaction constant values off by an order of magnitude. We’ve been capable to calculate pka for carboxylic acids and pkb values for amino groups accurately […]
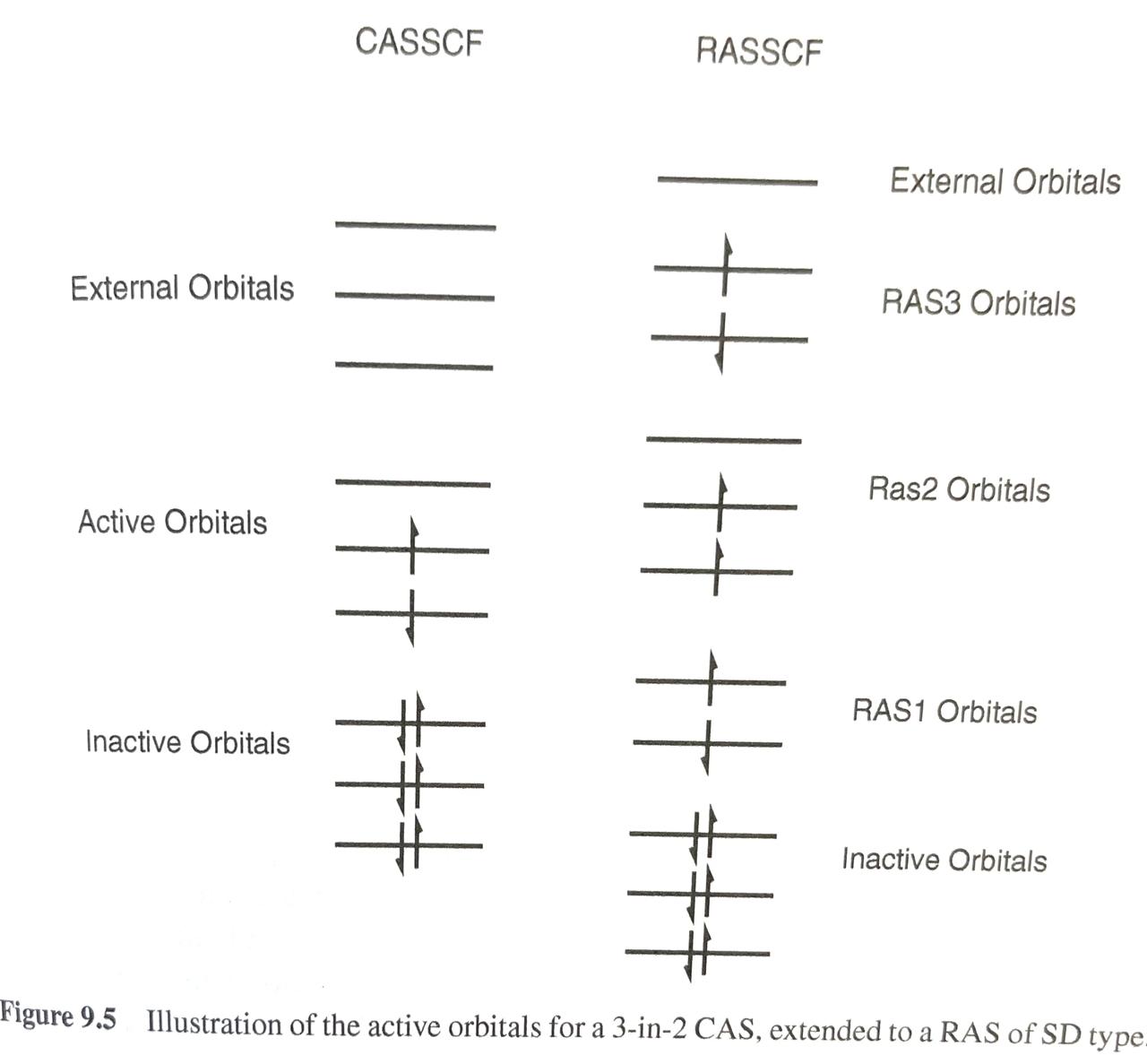
There are basically two kinds of electron correlation: static and dynamic. The former is mostly related to the lack of ab initio methods, namely Hartree-Fock, to fully describe the state of a polyelectronic system with a single electron configuration (described in turn by a single Slater determinant), therefore the use of Multiconfigurational methods ensures a […]
I had the honor to be invited by Dr. Michael Rauch from Schrödinger Inc. to work one of their online courses for learning how to use Materials Science (MS) Maestro. From their various options, I chose the Surface Chemistry course because it was an opportunity to brush on my knowledge about calculations on extended phases, […]
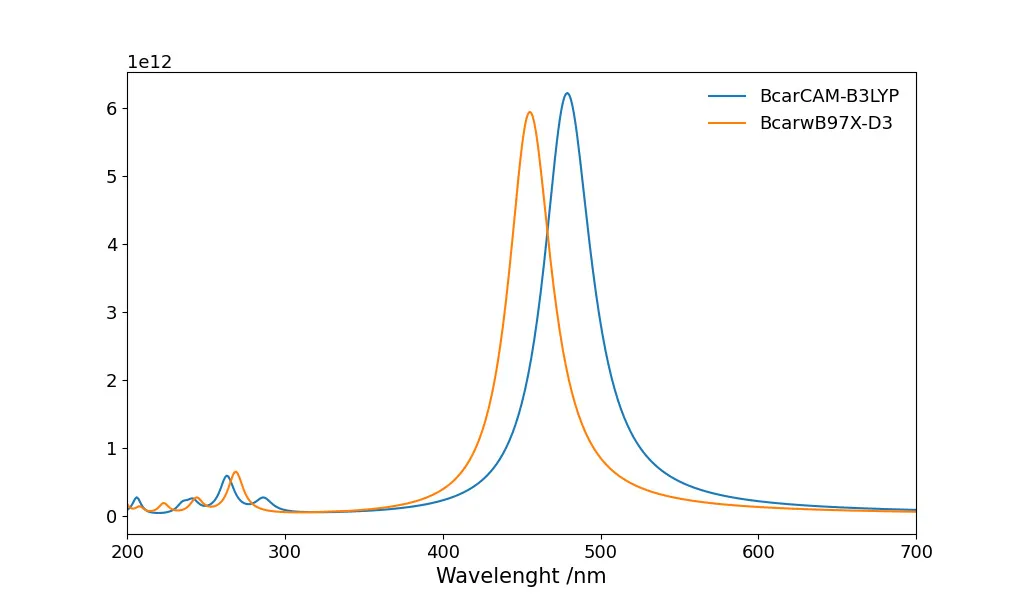
This new version of UVeh-Vis contains modifications of the graphical properties of the output plot: A UVeh-Vis comparison for a carotene molecule calculated at two different levels of theory using a lorentzian adjustment (Matplotlib) Any issue can be uploaded to the repository issues (https://github.com/joaquinbarroso/UVeh-Vis/issues) and extra features coming soon.If our code is useful for your …
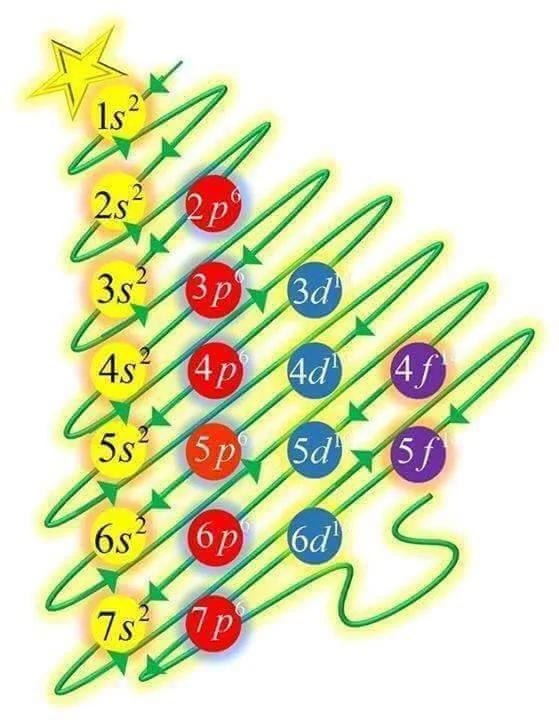
One of the first things I remember learning about chemistry in middle school (back in the early nineteen nineties) was how to write the electronic configuration of atoms using a sort of arcane code of numbers and letters in a weird progression hard to be rationalized, if not for a diagonal scheme that showed the […]
Last week I wasn’t invited by CNN to participate in a webinar to talk about who might win this year‘s Nobel prize for Chemistry. Aside from addressing who our favorite candidates were we also talked about how polymathy seems to be a common thread in a creative job such as being a scientist. Listen to […]
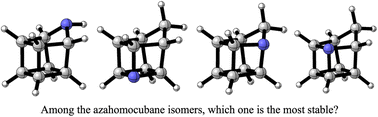
Cubane (C8H8) and homocubane (C9H10) are very strained molecules. Substitution of a carbon atom for a nitrogen one to reduce the tension in homocubane has been elusive until recently (Fahrenhorst-Jones et al. 2022). In contrast to cubane, substitution of C by N can occur at four different positions, so the obvious question is which one […]
 research.io
research.ioSign up to keep scrolling
Create your feed subscriptions, save articles, keep scrolling.