Scilife
Most companies think quality is a cost. In reality, poor quality is the actual expense. In the pharmaceutical industry, poor quality comes with a significant and measurable cost . It can range between 15% and 40% of total sales revenue , as found by different studies . That means a significant portion of your company’s resources may already be lost to deviations, rework, batch failures, and ineff…

We’re wrapping up Q1 with a strong set of updates, bringing more speed, structure, and traceability across your quality processes.

The temptation to think of EU MDR certification as a milestone is strong. Something you achieve, check off your list, and move on from. But in practice, it’s part of the ongoing lifecycle management of your device.
If you’re here, chances are you’re considering, or already actively searching for, a knowledgeable life sciences consultant to help pave the way for your organization’s future growth. Maybe you need support achieving ISO 13485 certification, obtaining your CE mark, or overcoming challenges with international market access for your medical device.
SharePoint has become an accidental QMS for many life sciences organizations. And look, we get it. It’s “free” (or at least already included in your Microsoft 365 subscription), it lets you build folder structures, and you can organize quality documents into whatever taxonomy or hierarchy makes sense to you. On the surface, it feels like a quick win.
Tech innovations are driving the rapid evolution of everything humans do, and auditing is no exception. Since 2020, remote or virtual audits have become commonplace, born out of necessity in a time when on-site audits were simply impossible. Due to the pressing circumstances, the transition from on-site to remote has been extraordinarily quick, and many auditees, as well as auditors, are still fa…
In the pharmaceutical industry, ensuring the safety and quality of medicines is a big responsibility. The U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are the two most influential regulatory agencies overseeing drug approval and compliance. Their inspections are rigorous, designed to ensure adherence to Good Manufacturing Practices (GMP), and Good Clinical Pract…
Introduction to Audits An audit is the verification, inspection, or examination of a process, product, or system to guarantee compliance with regulatory or organizational requirements. The scope of an audit can apply to an entire organization or be limited to a specific function, step, or process. Reasons for Audits - An internal audit or a first-party audit is performed to measure an organizatio…
Internal audits can strengthen the quality management system (QMS) of your organization by improving sub-processes by identifying open issues and opportunities. In addition, internal audits can be more informal than external audits, so that your organization can simulate an external audit in a less stressful setting. Although various regulations mandate internal audit compliance, it also has many…
FDA QMSR is now in effect—are you ready? Learn what the new regulation requires beyond ISO 13485:2016 and how to close gaps in risk management, documentation, and inspection readiness. A practical session to build a compliant, audit-ready QMS. EMEA Office Louizalaan 489 1050 Brussels Belgium
Pharma audits are not routine checks. They are moments of truth. Whether it’s an internal audit, a supplier audit, a customer audit, or a regulatory inspection, the question is always the same: Can you prove, with evidence, that your Quality Management System (QMS) -aka Pharmaceutical Quality System (PQS)- is under control?
Most people know the name of a medicine, but not the part that actually makes it work. That part is the active pharmaceutical ingredient (API), and in pharmaceutical manufacturing, its quality is everything.
Quality management systems (QMS) refer to the set of policies, procedures, processes, and resources that a company or organization puts in place to ensure that their products or services consistently meet or exceed customer requirements and expectations. A QMS is designed to help organizations achieve their quality objectives by documenting best practices, focusing on customer satisfaction, conti…
Top 11 FAQs about GMP Guidelines for Quality Management Systems The GMP guidelines are mandatory in most countries for pharmaceuticals, medical devices, and food industries. Companies that fail to comply with these guidelines can face severe regulatory consequences. For this reason, GMP certification is an essential prerequisite to be in the life science industry. If you are working on building a…
In my experience working with quality teams, GxP compliance in pharma is often perceived as a burden. However, it's a framework that protects what matters most to our industry: patients, product quality, and regulators' trust. Every stage of the journey, from the first experiments in the lab to commercial manufacturing, global distribution, and post-market monitoring, is shaped by GxP standards. …
GxP-compliance Software as a Service (SaaS) is becoming increasingly popular owing to its flexibility and ease of use. GxP compliance software on the cloud uses the internet to conveniently make software available for direct use to its clients. It can get clients to get up and running quickly without going through lengthy installation processes, not to mention that it provides many benefits to th…
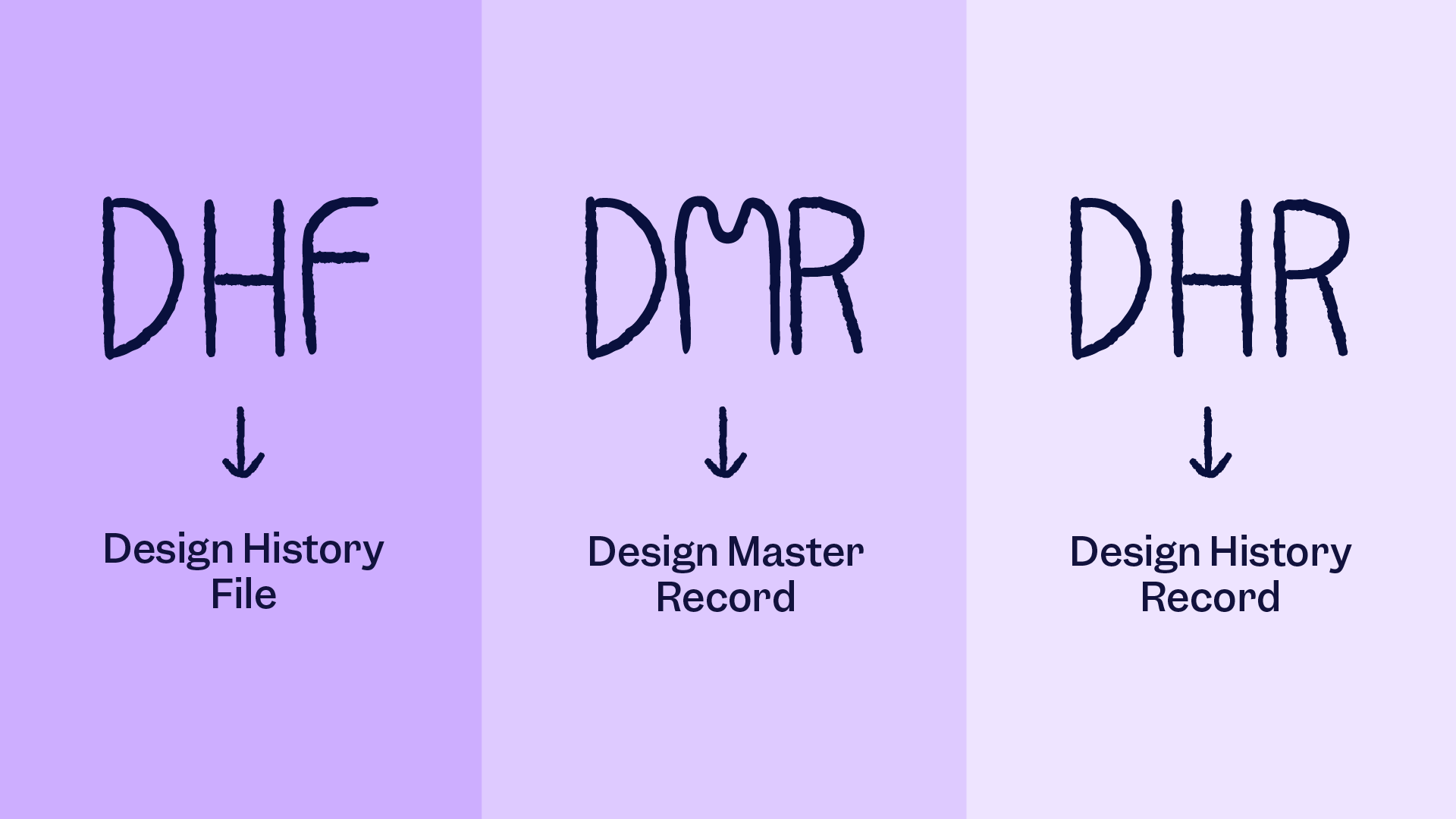
In the medical device industry, there are tons of abbreviations. And as a medical device professional in the US working with Quality Management Systems Regulations (QMSR), or 21 CFR Part 820 as it’s technically known, you can easily get lost in this acronym ocean! Until recently, the most important (and most confusing) abbreviations to understand were: DHF, DMR, DHR. These three abbreviations hav…
The FDA is transitioning from Computer System Validation (CSV) to Computer Software Assurance (CSA) for computers and automated data processing systems used as part of medical device production or medical device quality systems. So, what is the difference? How can your life science organization utilize either approach? And what are the benefits of transitioning to CSA? Both approaches play a simi…
In the era of Quality 4.0, cloud cybersecurity has become a crucial concern for businesses. Many companies are exploring the impact of switching from on-premise Quality Management Systems (QMS) to cloud-based QMS. It is important to understand the differences between these two types of QMS. In this article, we will begin by identifying the key differences between a cloud-based QMS and an on-premi…
 research.io
research.ioSign up to keep scrolling
Create your feed subscriptions, save articles, keep scrolling.