medical-devices


Scientists at Penn State have developed soft, stretchy electronic implants that could help treat people with dangerous high blood pressure that does not respond to medication. The new devices gently stimulate a major artery in the neck using tiny electrical signals. In animal tests, the implants lowered blood pressure while causing much less tissue damage […] The post This stretchy implant could …

Researchers at Penn State have developed a new class of 3D-printed bioelectronics made of soft, stretchy materials, as well as an adhesive component that helps the device painlessly stick to biological tissue. The team reported that their new design, which they call CaroFlex and tested in a rodent model, relieved hypertension while causing much less damage to surrounding tissue.
Bench to Bedside APEX is a global competition for student-founded companies developing medical devices, diagnostics, and health technologies. Teams present their business plans and innovations to a panel of judges representing clinical, engineering, and investment expertise. The competition emphasizes device-focused innovation with the potential to improve patient outcomes, streamline care, and a…

The medical device giant Medtronic won approval for its Altaviva implant, a tibial neurostimulator, in September. The device, roughly half the length of a stick of chewing gum (43.7 × 15.7 × 4.5 mm, 7.5 g), recently won a 2026 Edison Award for health innovation. Most recently, Medtronic recently enrolled its first patient in the… The post Medtronic launches five-year post-approval study for Altav…

Bedfont® Scientific Limited, an innovative MedTech company that specialises in designing and manufacturing medical breath analysis devices, is thrilled to have been shortlisted in the Export Achievement category at the Science and Engineering Health Technologies Alliance (SEHTA) Healthcare Business Awards 2026.

Developing AI-guided, patient-operated home ultrasound probes that can produce a reliable medical image. The post Beyond the Clinic: A Blueprint For Developing Reliable, Edge AI-Enabled Medical Devices appeared first on Semiconductor Engineering .

Researchers from USC and Caltech aim to develop a tiny sensor and drug delivery system, implanted near the eye, with dry eye disease as its first target. The post USC-led team receives funding to build next-generation medical device that uses tears to monitor health appeared first on USC .


Nervonik, Inc., a clinical-stage medical device company developing next-generation peripheral nerve stimulation (PNS) technologies, today announced the closing of an oversubscribed $52.5 million Series B financing.
the Ontogen Medtech Biomedical Innovation Scholarship is intended to support promising biomedical engineering students who are passionate about building real-world medical devices and contributing to patient impact—especially students pursuing hands-on product development, manufacturing, and commercialization pathways. Applications due August 31


Cala(r), the bioelectronic medicine leader, today announced U.S. Food and Drug Administration (FDA) clearance of the Cala kIQ(r) Plus system, the next-generation of its wearable neurostimulation device for action hand tremor in essential tremor (ET) and Parkinson's disease (PD).

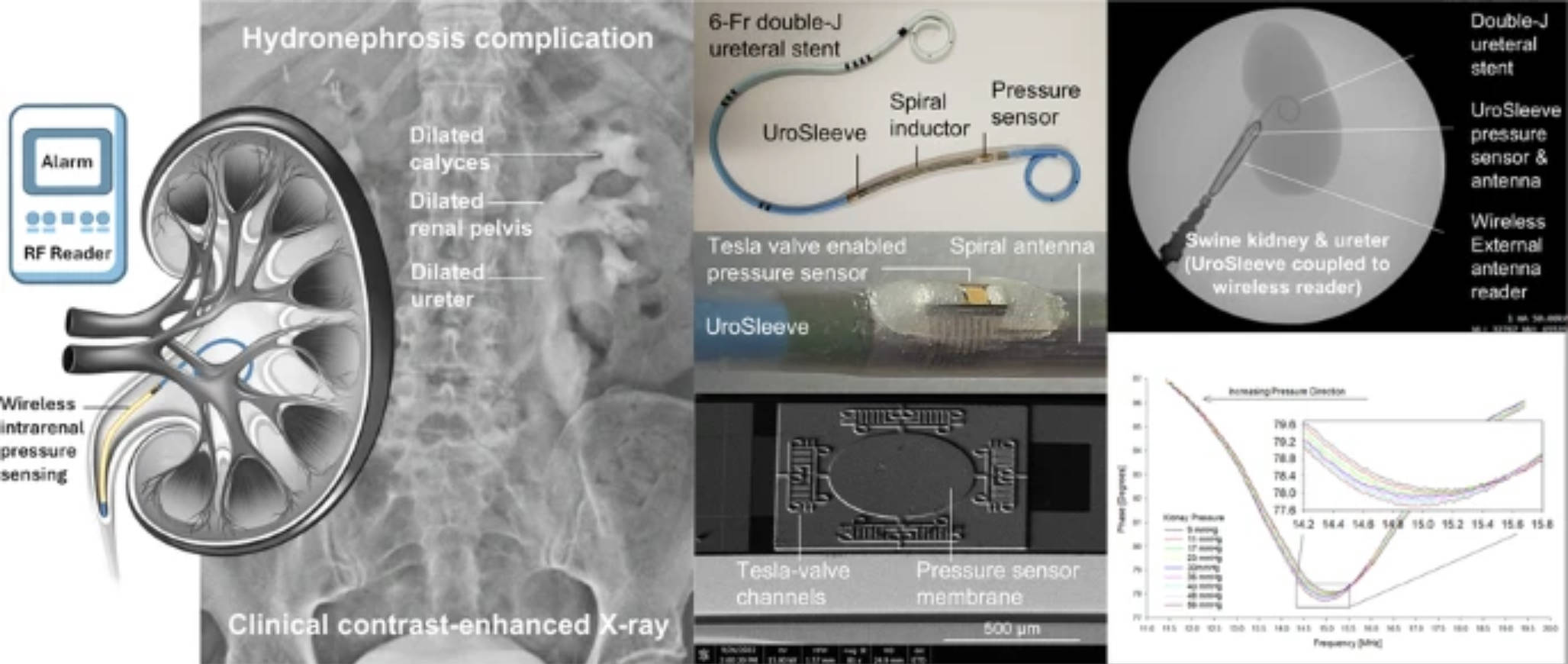
When a ureteral stent fails, pressure can quietly build inside the kidney long before clinical symptoms. A new device called UroSleeve aims to enable earlier detection by turning a standard stent into a passive wireless pressure monitor via a modular sleeve. Designed as a modular sleeve rather than a redesigned implant, it fits around conventional stents and tracks intrarenal pressure through res…

A team of engineers, chemists and physicians at UNC-Chapel Hill is developing a breakthrough membrane technology that could shrink today’s refrigerator-sized dialysis machines into a wearable, smartphone-sized artificial kidney—potentially transforming care for hundreds of millions of patients worldwide. The post A Smartphone Moment for Kidney Dialysis appeared first on Department of Chemistry .

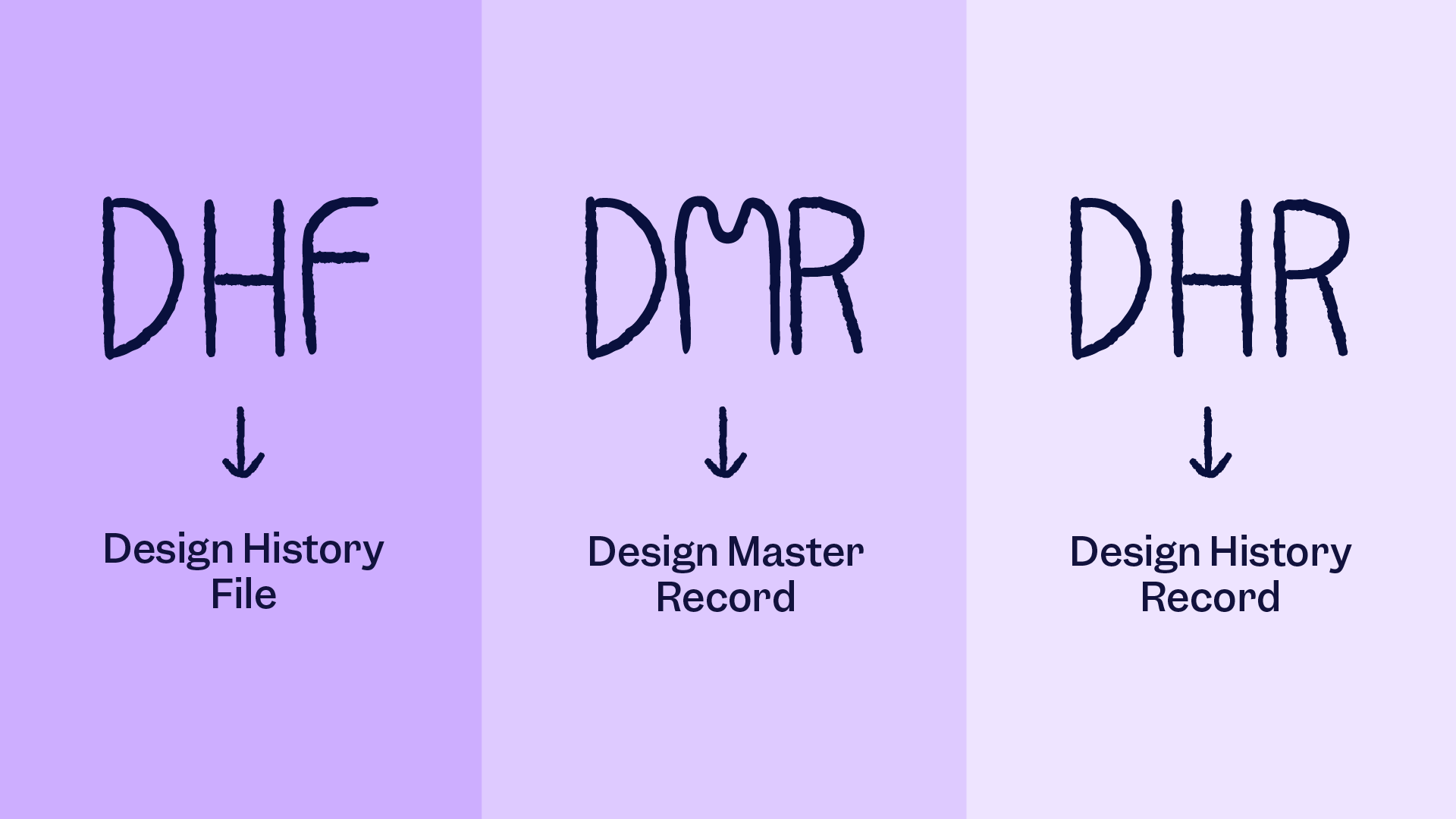
In the medical device industry, there are tons of abbreviations. And as a medical device professional in the US working with Quality Management Systems Regulations (QMSR), or 21 CFR Part 820 as it’s technically known, you can easily get lost in this acronym ocean! Until recently, the most important (and most confusing) abbreviations to understand were: DHF, DMR, DHR. These three abbreviations hav…

Developing and bringing a medical device to market is a scary process. Not only do you have to come up with a great idea or design improvement and develop a working prototype, but you must also ensure the device is safe and performs as intended in any indicated situation. Potentially the most daunting task of medical device development is the regulatory aspect. Ensuring regulatory compliance can …

If you work in medical devices in the EU, the question is no longer what EUDAMED is, but whether your data and ownership model will survive mandatory use.

New technology could make diabetes management more accessible to those who need it most. The post Microneedles enable 98% smaller, cheaper artificial pancreas appeared first on Advanced Science News .

Developing and manufacturing a stable and safe medical device is difficult for even the most experienced manufacturers. How can you ensure you manufacture highly predictable medical devices that offer consistent performance with minimal variation? The answer is process validation. Process validation is the framework around your medical device manufacturing process that ensures your device is inst…

Many medtech teams are laser-focused on building the perfect product—sleek prototypes, groundbreaking technology, elegant user interfaces. But even the most brilliant design can stall if compliance is left as an afterthought. Whether you're preparing for CE marking, FDA clearance, or MDR compliance, the demands on your quality and product teams are rising. But many still rely on spreadsheets, dis…

Risk management is a vital part of medical device development and lifecycle processes. Most regulatory agencies, including central authorities such as the European Union and the Food and Drug Administration (FDA), include risk-based methods in their internal processes for medical device and manufacturer evaluations. They also require medical device manufacturers to apply risk management processes…
 research.io
research.ioSign up to keep scrolling
Create your feed subscriptions, save articles, keep scrolling.