regulatory-health


In our recent blog examining the FDA’s move toward a one pivotal trial approval pathway, we explored what this shift means for clinical strategy, regulatory engagement, and evidentiary rigor. As development programs concentrate approval decisions into a single adequate and well-controlled study, the implications extend well beyond protocol design and statistical planning. If one study carries th…


In February 2026, the Food and Drug Administration (FDA) outlined a meaningful evolution in the Agency’s evidentiary approach to drug approval: establishing one adequate and well-controlled trial, supported by confirmatory evidence, as the default basis for marketing authorization. This position was articulated in The New England Journal of Medicine in “One Pivotal Trial, the New... The post The …


Gaining regulatory approval for drugs targeting conditions that cannot ethically be studied in humans presents a unique challenge for drug developers. Without traditional clinical trial pathways, teams must rely on alternative methods to demonstrate efficacy and support approval. This webinar will explore how modeling and simulation are used to meet the requirements of the FDA […] The post Naviga…

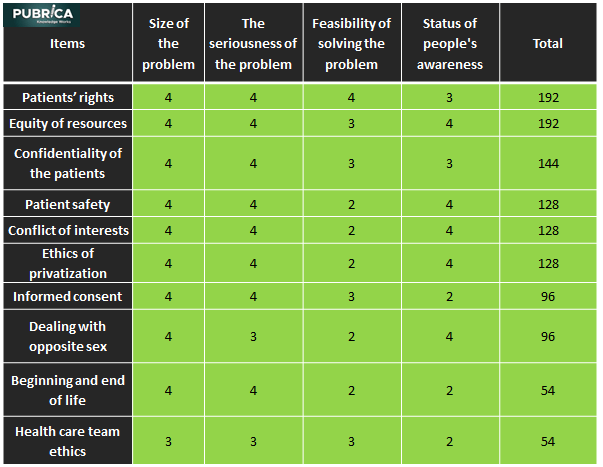
In Brief Biotechnological advancements in medical devices can intensify risks and introduce new types of risk, despite their potential to greatly benefit health. Therapeutic products control […] The post How to overcome regulatory and ethical challenges regarding medical device? appeared first on Pubrica .