regulatory-compliance



Developing and bringing a medical device to market is a scary process. Not only do you have to come up with a great idea or design improvement and develop a working prototype, but you must also ensure the device is safe and performs as intended in any indicated situation. Potentially the most daunting task of medical device development is the regulatory aspect. Ensuring regulatory compliance can …

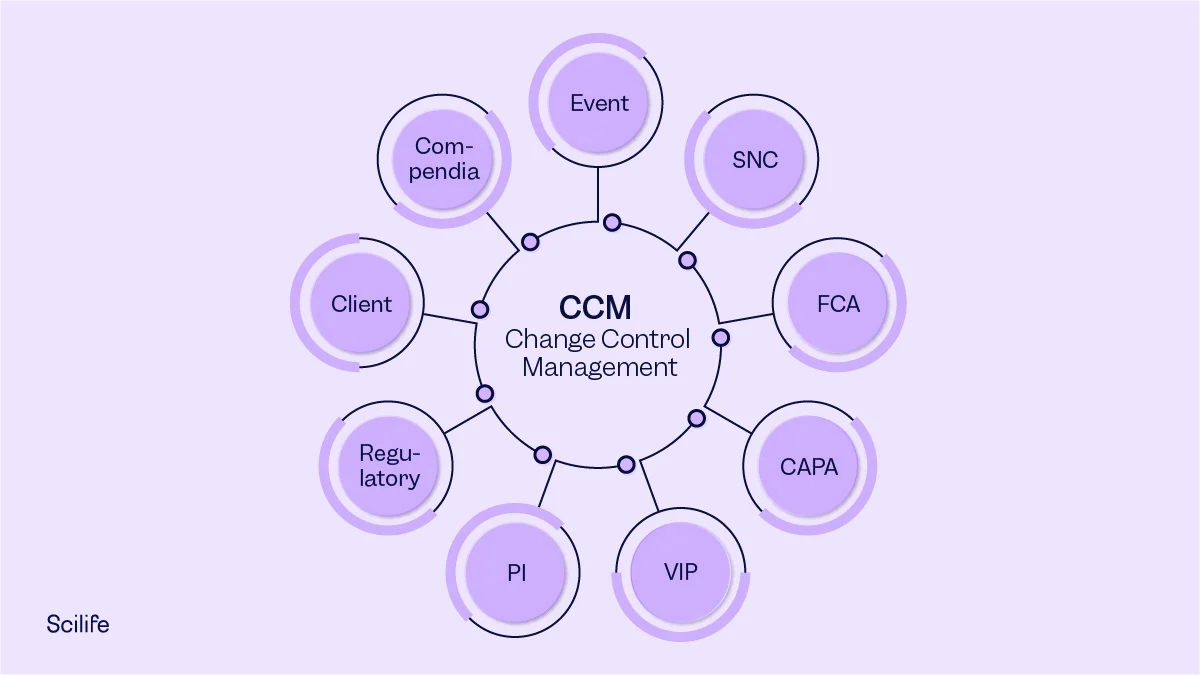
The requirements of life sciences companies (including in the pharma and medical device space) inevitably change over time. Sometimes, a change is due to regulatory requirements; sometimes, customer needs change. Keep reading if you want to learn more about what change is, how to handle change, and what steps you need to take for a successful change control process. What is Change Control in Phar…

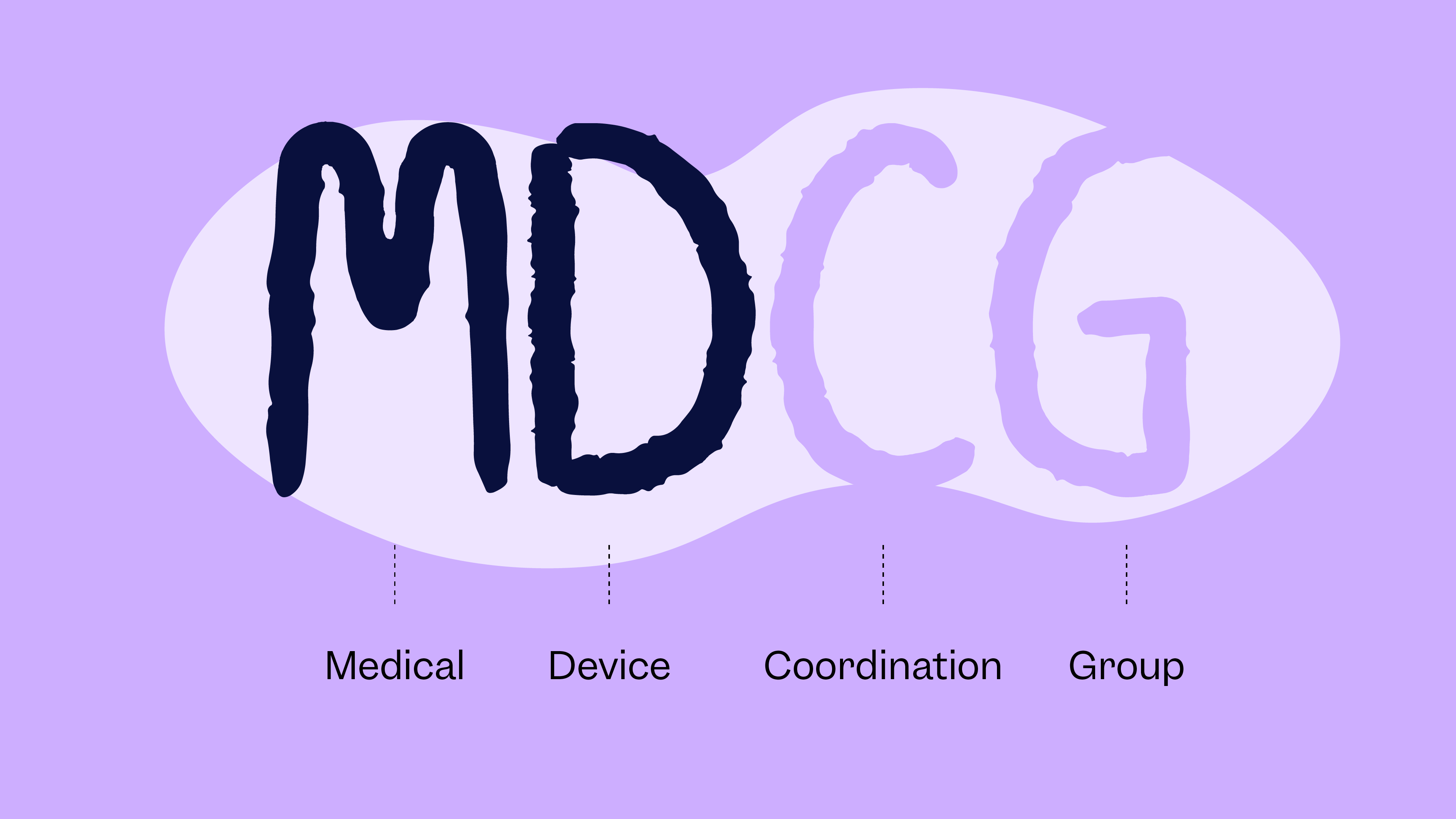
A fundamental part of safeguarding patients in Europe comes from ensuring that medical devices and in vitro diagnostics (IVDs) meet consistent, high regulatory standards. Under the EU Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR), those standards are shaped by detailed legislation. That’s where MDCG guidance—issued by the Medical Device Coordination Group—comes in. It …

We all know that pharma companies are duty-bound to control the quality of the drugs they produce—it's paramount for protecting patient safety. One critical part of how they do this is by employing a Corrective and Preventive Action (CAPA) system. CAPA is a systematic approach for identifying quality issues with products and then fixing those issues so they don’t happen again. Pharma companies (l…

If you’ve ever been through an audit in the life sciences industry, you know how important Corrective and Preventive Actions (CAPAs) are. Regulatory bodies expect you to have a strong CAPA process in place to uphold product quality and patient safety. But let’s be honest, CAPA management can be one of the most frustrating and time-consuming parts of managing a quality system. I’ve seen companies …

Plan medical device compliance smarter with a clear cost breakdown, testing and validation strategies, common overspending pitfalls, and a practical checklist to reduce regulatory expenses.

You’ve finally received your 510(k) clearance and feel ready to start commercialization in the US market. But how do you go about importing medical devices into the US? Importation is the final hurdle in an often year-long process to obtain regulatory clearance/approval in the US market. Without being able to import your medical devices, all the hard work that came before becomes irrelevant. In t…

Root cause investigation (RCI) is a problem-solving approach for gathering significant information and data regarding a particular problem or quality deviation. The primary goal of RCI is to identify the root cause of any existing or potential nonconformities and provide suitable solutions. The investigation process includes data collection, interviews, observations, and review. In the highly reg…

Having the FDA knocking on your door for a medical device inspection is scary! However, the management of FDA inspections for medical devices is a critical aspect of ensuring compliance with regulatory standards and maintaining the integrity of the medical device industry. The US Food and Drug Administration (FDA) is responsible for safeguarding public health by regulating medical devices and med…

What is EU GMP Annex 11? EU GMP rules, also known as the ‘EudraLex rules’ govern the medicinal products in the European Union. The EU GMP rules are split into three different parts plus about 20 annexes. ‘The EU GMP Annex 11’ is one of the supplementary documents of EU GMP rules. The growing use of computerized systems in life sciences, alongside their intricate structures, has led to the necessi…

Good Manufacturing Practices (GMPs) have been the cornerstone of quality assurance in pharmaceuticals and medical devices for decades. These guidelines ensure that every product, from medicines to medical devices, is consistently produced and controlled to the highest quality and safety levels. GMP standards cover all aspects of production, from starting materials, premises, quality assurance, te…

Ensure your medical device meets regulatory standards with our expert Clinical Evaluation Reports (CER) services. We provide comprehensive support from data collection to evidence appraisal, helping you achieve compliance efficiently. Enhancing Compliance and Market Readiness of Medical Devices Through Expert CER, Post-Market Surveillance, and Data Collection Services The medical device industry …

Keeping track of medical devices is paramount to ensure the safety of patients. Traceability allows manufacturers to identify, trace, and control medical devices and is necessary for several reasons, chief among them regulatory compliance. Regulatory bodies such as the Food and Drug Administration in the United States and the European Medicines Agency (EMA) in the European Union mandate strict re…

The United States Food and Drug Administration (FDA) is the regulatory body responsible for the safety and efficacy of medical devices in the United States. The 510(k) clearance is their classic premarket clearance for Class II medical devices that most of us are probably familiar with. The 510(k) demonstrates that devices are substantially equivalent to similar devices on the market, clearing th…

Cell and gene therapies hold great promise for improved health outcomes. Now is the time to advance life-saving research to and through the clinical setting. Critical to this is the aspect of raw materials standardization and regulatory compliance, in which it would serve you well to identify and address key considerations early on. To help The post Your Starting Point For Cell & Gene Therapy Reg…